Regulatory trigger values: Appropriateness, assumptions and gaps in science
The variation that exists for log KOW trigger values for potential bioconcentration among regulatory guidelines suggests there is a level of uncertainty regarding what constitutes an appropriate value. In terms of ERA of chemicals, the TGD (EC, 2003) has been a common frame of reference for environmental risk assessors wanting to understand the relevance of log KOW as an indicator of potential to bioconcentrate. Accordingly, the most important and widely accepted indication of bioaccumulation potential is a high value of log KOW (EC, 2003). It is also maintained that for certain substances such as those that ionise in water, log KOW values may not be suitable to calculate a BCF value. Therefore, the TGD explicitly acknowledged that the relationship between log KOW and BCF for neutral organic compounds is predictive, whereas for ionisable substances the pH of the environment will influence the relevant quantities of neutral and dissociated species present. This has obvious implications on partitioning behaviour.
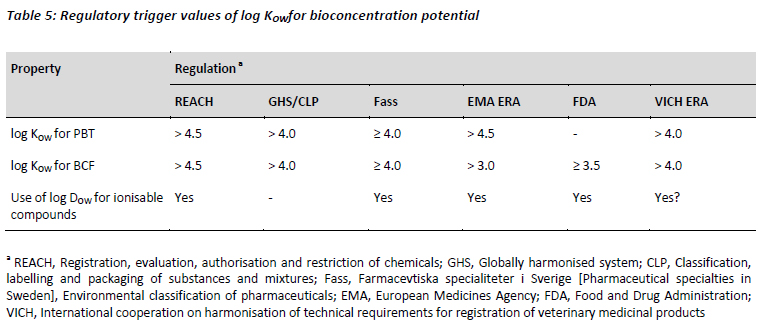
The distinction between neutral and ionised compounds in terms of their influence on bioconcentration potential is reiterated in the EU by the present REACH guidance and by other regulators (Table 5).
For REACH guidelines the relative extent to which an ionisable substance is likely to be dissociated in the environment (pH 5 - 9) can have a marked effect on its physical-chemical properties, especially the KOW and water solubility, which in turn affect fate and behaviour (ECHA, 2008b). Moreover, the value for the dissociated molecule determined around a pH of 7 (sometimes referred to as DOW) is considered more realistic for PBT and chemical safety assessment. However, REACH differs from its predecessor in that it considers the appropriate trigger for bioconcentration potential to be log KOW . 4.5, rather than 3.0. It seems likely that this latter value was adopted by the EMA ERA guidelines which, when finalised in 2006, required that a bioconcentration study be considered for pharmaceuticals exceeding a KOW of 1,000 (EMEA, 2006). It is not evident whether the ionisable nature of most APIs was a consideration for these guidelines. However, more recently the EMA have recognised this property of APIs and clarified that for ionisable compounds log DOW should be determined as a function of pH covering an environmentally relevant pH-range. Unlike REACH, the trigger value for BCF evaluation remains at log KOW . 3.0 (EMA, 2010). A different trigger value is considered by the Swedish Classification of Pharmaceuticals guidelines which, taking its cue from the CLP guidelines, states that bioconcentration potential may be indicated by log KOW . 4. The use of log DOW at pH 7 as an indicator of bioconcentration for complex ionic molecules is explicitly accepted by these guidelines. In the USA, the FDA Environmental Assessment guidelines also acknowledge that for substances which dissociate in water, the KOW may need to be evaluated at pH 5, 7 and 9. Once again, the trigger value differs from those in Europe, whereby a log KOW . 3.5 indicates that chronic toxicity testing may be required.
Overall, whilst there are differences between the regulatory log KOW values (ranging 3.0 - 4.5) for bioconcentration potential, there is consensus that evaluation of the distribution coefficient (log DOW) is appropriate for chemicals that dissociate in water at environmentally relevant pH 5 to 9. For pharmaceuticals with ionisable groups, it follows axiomatically that the charged state of these groups will vary according to the pKa and environmental pH; this in turn may influence water solubility and partitioning behaviour. While there is significant support for this observation in the scientific literature (Avdeef, 1996; Kah and Brown, 2008; Rayne and Forest, 2010), the scientific basis for asserting log DOW is more appropriate for predicting bioconcentration compared with the partitioning of the neutral form (log KOW) remains largely untested.